| Fluorine | |
| Atomic number | 9 |
| Appearance of a simple substance | Fluorine in a vessel Pale yellow gas, extremely chemically active. Very poisonous. |
| Properties of the atom | |
| Atomic mass (molar mass) | 18.998403 a. e.m. (/mol) |
| Atomic radius | 71 pm |
| Ionization energy (first electron) | 1680.0 (17.41) kJ/mol () |
| Electronic configuration | [He] 2s2 2p5 |
| Chemical properties | |
| Covalent radius | 72 pm |
| Ion radius | (-1e)133 pm |
| Electronegativity (Pauling) | 3,98 |
| Electrode potential | 0 |
| Oxidation states | −1 |
| Thermodynamic properties of a simple substance | |
| Density | (at −189 °C)1.108 /³ |
| Molar heat capacity | 31.34 /(mol) |
| Thermal conductivity | 0,028 /(·) |
| Melting temperature | 53,53 |
| Heat of Melting | (FF) 0.51 kJ/mol |
| Boiling temperature | 85,01 |
| Heat of vaporization | 6.54 (FF) kJ/mol |
| Molar volume | 17.1 ³/mol |
| Crystal lattice of a simple substance | |
| Lattice structure | monoclinic |
| Lattice parameters | 5.50 b=3.28 c=7.28 β=90.0 |
| c/a ratio | — |
| Debye temperature | n/a |
| F | 9 |
| 18,9984 | |
| [He]2s22p5 | |
| Fluorine | |
Fluorine
- an element of the main subgroup of the seventh group, the second period of Mendeleev’s periodic system of chemical elements, with atomic number
9
.
Identified by the symbol F
Fluorum.
Fluorine is an extremely reactive non-metal and the strongest oxidizing agent; it is the lightest element from the halogen group. The simple substance fluorine
(CAS number: 7782-41-4) under normal conditions is a pale yellow diatomic gas with a pungent odor reminiscent of ozone or chlorine (formula F2). Very poisonous.
Story
Diagram of a fluorine atom
The first fluorine compound, fluorite (fluorspar) CaF2, was described at the end of the 15th century under the name “fluor”. In 1771, Karl Scheele obtained hydrofluoric acid.
As one of the atoms of hydrofluoric acid, the element fluorine was predicted in 1810, and isolated in its free form only seventy-six years later by Henri Moissan in 1886 by electrolysis of liquid anhydrous hydrogen fluoride containing an admixture of acidic potassium fluoride KHF2.
origin of name
The name “fluorine” (φθόρος - destruction), proposed by Andre Ampère in 1810, is used in Russian and some other languages; Many countries have adopted names derived from the Latin "Fluorum" (which in turn comes from fluere
- “flow”, according to the property of the fluorine compound, fluorite (CaF2), lowers the melting point of the ore and increases the fluidity of the melt).
Lack of fluoride in the body
Since the main source of fluoride for humans is water, it is necessary to ensure that it contains this element in sufficient quantity.
A lack of fluoride in the body can be expressed by the following symptoms:
- split ends, brittleness, dryness and hair loss:
- development of caries, increased sensitivity of enamel, crumbling of teeth, increased abrasion, erosion and necrosis of the gums;
- frequent and repeated fractures, cracks and difficulties with bone healing, which may be a sign of osteoporosis;
- susceptibility to colds and viral diseases due to weakened immunity.
The most effective way to eliminate fluoride deficiency in the body is to change the source of drinking water. It is necessary to choose bottled water containing this element. If there is a significant fluoride deficiency, confirmed by tests, appropriate therapy is prescribed. You can improve the condition of your teeth with the help of fluoride-containing toothpaste, which should be selected by a professional dentist.
The method of replenishing the mineral is selected based on symptoms and the presence or absence of concomitant diseases and contraindications. When calculating the dosage of a particular drug to replenish fluoride reserves, you should avoid exceeding the dose, since this element is contained in some foods, water and vitamins. For this reason, it is better to consult a doctor to select the necessary remedy and its dosage.
Distribution in nature
The fluorine content in atomic percentages in nature is shown in the table:
| An object | Content |
| The soil | 0,02 |
| River waters | 0,00002 |
| Ocean waters | 0,0001 |
| Human teeth | 0,01 |
In nature, significant accumulations of fluorine are found only in the mineral fluorite.
Lentils and onions are relatively rich in fluoride to some extent.
The fluorine content in the soil is due to volcanic gases, due to the fact that their composition usually includes a large amount of hydrogen fluoride.
Fluoridation of teeth in dental
Deep fluoridation is carried out using a modern APF system.
Fluoridation of teeth is a procedure for the prevention and strengthening of the superficial and deep layers of tooth tissue. Teeth require two substances - calcium and fluoride, but they constantly displace each other from the surface of the enamel. Therefore, a new preparation APF gel has appeared, based on nanotechnology, which is capable of saturating the deep layers of enamel with calcium, and the upper layers with fluoride. The fluoridation procedure for teeth using the APF system takes 10-20 minutes. To consolidate the effect of the procedure, a product for further home care is given to your home. This procedure allows not only to protect teeth from caries, but also to restore enamel with caries already present on the surface.
Isotopic composition
Fluorine is a monoisotopic element, since only one stable isotope of fluorine, 19F, exists in nature. Another 17 radioactive isotopes of fluorine are known with a mass number from 14 to 31, and one nuclear isomer - 18Fm. The longest-lived radioactive isotope of fluorine is 18F, with a half-life of 109.771 minutes, an important source of positrons used in positron emission tomography.
Nuclear properties of fluorine isotopes
| Isotope | Relative mass, a.m.u. | Half life | Type of decay | Nuclear spin | Nuclear magnetic moment |
| 17F | 17,0020952 | 64.5 s | β+ decay in 17O | 5/2 | 4.722 |
| 18F | 18,000938 | 1.83 hours | β+ decay in 18O | 1 | |
| 19F | 18,99840322 | Stable | — | 1/2 | 2.629 |
| 20F | 19,9999813 | 11 s | β− decay in 20Ne | 2 | 2.094 |
| 21F | 20,999949 | 4.2 s | β− decay in 21Ne | 5/2 | |
| 22F | 22,00300 | 4.23 s | β− decay in 22Ne | 4 | |
| 23F | 23,00357 | 2.2 s | β− decay in 23Ne | 5/2 |
Magnetic properties of nuclei
The nuclei of the 19F isotope have half-integer spin, so these nuclei can be used for NMR studies of molecules. The 19F NMR spectra are quite characteristic of organofluorine compounds.
Physiological action
The adult human body contains about 2.6 g of F. Almost all of it is included in the composition of bones and teeth. This is the main value of this microelement - it strengthens our bones and teeth. But what about calcium?
After all, it is common knowledge that calcium is the building cement that gives strength to bone tissue. Yes it is. But then what does fluorine have to do with it?
And fluoride facilitates the deposition of calcium in bone tissue. Together with calcium, another important element for bones and teeth, phosphorus, is deposited. These two macroelements, calcium and phosphorus, are found in bones and teeth in the form of calcium phosphate, or more precisely, its variety, apatite.
The structural basis of apatite molecules has the form Ca10(PO4)6. Chlorine, fluorine, and hydroxide ions (OH) can be attached to it.
In this regard, fluorapatites, chlorapatites, and hydroxyapatites are distinguished. The most durable of them is fluorapatite. Fluorine makes the crystal structure of apatites more ordered and prevents their destruction under the influence of an acidic environment. It can initially attach to the apatite molecule, or, as the most active chemical element, displace hydroxide ions from it.
As a result, fluorapatites enhance the mineralization of bones and teeth, and thereby give them strength. F prevents bone demineralization associated with changes in pH (acidity), infection, metabolic disorders, and other damaging factors. Thus, it prevents the development of such common negative phenomena as dental caries and bone osteoporosis.
Dental caries is a complex pathological process. An important role in its development is played by the acid-forming microflora of the oral cavity. Under its action, glucose is broken down into organic acids.
As a result, the pH value in the oral cavity decreases, and acidity, accordingly, increases. At the same time, stains form on the tooth enamel, and foci of destruction form directly under the stains.
It stimulates the processes of remineralization, restoration in damaged enamel and in the deeper tissues of the tooth, dentin, and thereby eliminates existing defects.
In bone tissue, it controls the proper deposition of calcium phosphate. Thanks to it, the risk of fractures is reduced, and the resulting fractures heal faster.
In childhood and adolescence, with the participation of fluoride, the bone skeleton is formed correctly.
It should be noted that bones and tooth enamel are not the leaders in fluoride content. There is more of it in dental cement. Then comes bone tissue, dental dentin, and only then tooth enamel. In addition to bones and teeth, F has a positive effect on the condition of nails and hair, stimulates their growth, and increases strength.
In addition, it is part of some tissue enzymes. With these enzymes, it promotes the synthesis of leukocytes and improves immunity. It has been established that the microelement inhibits the growth of acid-forming bacterial microflora of the oral cavity. Thanks to this, it has an additional effect on dental caries.
Not only the formation of leukocytes, but also erythrocytes and hemoglobin is stimulated by fluoride. This is due to the fact that it is involved in iron metabolism. Thanks to this, it has a positive effect on the state of hematopoiesis and the cardiovascular system. With its participation, the binding and removal of salts of heavy metals, radionuclides, and other toxic compounds occurs.
Daily requirement
The need for F depends on age and some other factors.
| Category | Daily value, mg |
| Children under 2 years old | 0,5-0,7 |
| Children 3-5 years old | 0,9 |
| Children 6-8 years old | 1,1 |
| Children 9-13 years old | 1,3 |
| Teenagers, boys and girls | 1,5 |
| Adults, men and women | 1,7-4 |
| Pregnant and nursing | 4 |
During illnesses and intense physical activity, the daily requirement for F may increase to 5 mg.
Causes and signs of deficiency
The main cause of fluoride deficiency is due to low levels of this element in drinking water in some regions. Because of this, plants and animals that serve as food sources for people living in these regions do not receive enough of it.
Indicators of fluoride content in water:
| Content in drinking water, mg/l | Meaning |
| Up to 0.3 | Very low |
| 0,3-0,7 | Low |
| 07-1,2 | Optimal, normal |
| 1,2-1,5 | Acceptable, but elevated |
| 1,5-2 | Above acceptable |
| 2-6 | High |
| 6-15 | Very high |
In some sources, fluoride content values are displayed in mmol/l. At the same time, its distribution on the territory of the Russian Federation is as follows.
Sometimes a person himself artificially reduces its amount in drinking water. Filters used for purification remove not only toxic impurities from water, but also fluorine. Along with the low content in water and soil, F deficiency can be caused by an unbalanced diet, low consumption of fish, seafood, and other foods rich in it.
Another reason is the systematic use of certain medications. The fact is that aluminum and magnesium impair its absorption. The same applies to medicines containing these elements. These are Panangin, Asparkam, Cardiomagnyl, used in cardiological practice, and not only.
Antacids, agents for reducing the acidity of gastric juice (Almagel, Maalox), contain both aluminum and magnesium in the form of hydroxides. In chronic diseases of the gastrointestinal tract (gastrointestinal tract), the absorption of many nutrients deteriorates, incl. and fluoride.
Its deficiency is manifested by dental caries and pathological fragility of bones against the background of osteoporosis. When the deficiency is severe, teeth fall out. Elderly and old people are especially susceptible to osteoporosis. Its deficiency in old age increases the risk of hip fracture and other bone fractures.
In childhood and adolescence, lack of fluoride is one of the causes of spinal curvature: scoliosis, increased kyphosis. In addition, patients with its deficiency often suffer from anemia. Their immunity is reduced, and because of this, their susceptibility to infections increases. Appearance deteriorates. Hair becomes brittle and falls out. The nail plates peel off.
Sources of income
We get 2/3 of the fluoride we need with water, and the remaining 1/3 with food. The leader in F content is green tea. There is a lot of it in fish, seafood, meat and nuts.
F content in 100 g of products:
| Product | Content, mg/100 g |
| Green tea | 10 |
| Tuna | 1,5 |
| Mackerel | 2 |
| Cod | 0,7 |
| Salmon | 0,5 |
| Flounder | 0,43 |
| Oysters | 0,4 |
| Walnuts | 0,65 |
| Mutton | 0,2 |
| Chicken | 0,15 |
| Milk | 0,1 |
| Radish | 0,85 |
| Buckwheat | 0,1 |
| Potato | 0,1 |
It should be noted that when cooking food, a certain amount of F goes into water. To reduce microelement losses, food should be cooked in a small volume of water, and it is advisable not to exceed the required time.
It is highly undesirable to use aluminum cookware. It is better not to boil the fish, but to bake it. Fish with bones is superior to fish fillets in terms of fluoride content. Canned fish are also rich in it.
Synthetic analogues
Fluoride-containing compounds are included in toothpastes. These pastes strengthen teeth and prevent tooth decay. F in toothpastes is represented by the following compounds:
| Compound | Characteristics |
| Tin fluoride | It has a pronounced remineralizing effect on tooth enamel. Helps whiten teeth enamel. However, under its influence, bleached enamel darkens after some time. Negatively affects the condition of the gums. Due to negative qualities, it is currently not used. |
| Monosodium phosphate | Fluoride is released slowly from this compound. Therefore it is ineffective. |
| Aminofluoride | Remineralization is maximum. The compound forms a film on the surface of the tooth enamel. From this film, fluoride penetrates the enamel and deeper tissues of the tooth. |
| Sodium fluoride | It quickly decomposes to form F ions, and therefore acts just as quickly. |
| Nicomethanol fluorohydrate (Fluorinol) | The ability to fix and penetrate into tooth enamel and dentin is 12 times greater than inorganic fluorine compounds. |
Thus, sodium fluoride (another name is sodium fluorate) is one of the most popular remedies.
It is used not only in the form of toothpastes, but also in the form of mouth rinses, tablets and solutions for oral administration (Vitaftor, Coreberon, Ossin), varnishes for application to tooth enamel (Profluoride Varnish, Bifluoride 12, Fluorlak).
Sensigel dental gel and Elgifluor toothpaste contain Fluorinol as an active substance.
In addition to these products, it is part of a number of dietary supplements and complex vitamin-mineral preparations. Sodium fluoride, sodium fluorsalicylate, and fluorosilicic acid are used to fluoridate water. These connections have a number of advantages:
- they are cheap
- dissolves well in water
- safe for health
- do not change the properties of water: they maintain its transparency and do not impart taste or smell to it.
Periodic fluoridation of tap water in low F regions has been shown to strengthen teeth and reduce the incidence of tooth decay.
Metabolism
Fluoride comes in the form of inorganic compounds with water and food. A certain amount of the microelement can enter through the respiratory tract and be absorbed through the skin. Fluoride entering the gastrointestinal tract is absorbed by simple or facilitated diffusion in the stomach and in the initial parts of the small intestine.
Fluoride absorption is maximum, approaching 100%. Once absorbed, it is carried by the blood to organs and tissues. Up to 99% of F is concentrated in bones and teeth, and only 1% in other tissues. Almost all F is excreted from the body in urine. Only a small amount can leave the body in sweat and feces.
Interaction with other substances
Fluoride metabolism is closely related to the metabolism of calcium and phosphorus - thanks to it, these macroelements are deposited in bones and teeth. However, in large doses it negatively affects calcium metabolism. Vit. A and E improve its absorption.
Aluminum and magnesium, on the contrary, worsen this process. The same applies to drugs containing aluminum and magnesium compounds. Fluoride improves iron absorption. It worsens the absorption of other halogens, bromine, iodine, chlorine.
Electronic structure
Application of the MO method for the F2 molecule
The electronic configuration of the fluorine atom is as follows: 1s22s22p5. Fluorine atoms in compounds can exhibit an oxidation state of −1. Positive oxidation states are not realized in compounds, since fluorine is the most electronegative element.
Quantum chemical term of the fluorine atom - ²P3/2
Molecule structure
From the point of view of molecular orbital theory, the structure of a diatomic fluorine molecule can be characterized by the following diagram. The molecule contains 4 bonding orbitals and 3 antibonding orbitals. Obviously, the bond order in the molecule is 1.
Chronic fluoride poisoning
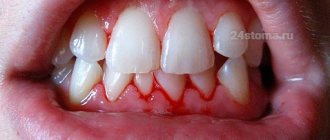
Most often, chronic fluoride poisoning is diagnosed in children and adolescents, but sometimes this condition also occurs in adults. In the case of a chronic form of intoxication, which occurs with prolonged exposure to this substance, a person experiences characteristic symptoms. At the initial stage - increased sensitivity and bleeding of the gums, brown spots on the surface of the teeth, caries, periodontal disease, soreness and swelling of the nose, ulcerative lesions of the nasal septum, dyskinesia of the intestines and stomach.
Later, diseases of the respiratory system appear, a person also begins to feel pain in the heart area, and his circulatory and hematopoietic processes are disrupted. With chronic intoxication, cholesterol metabolism malfunctions, stomach ulcers and gastritis develop. If excess fluoride is not removed from the body, over time it can negatively affect the condition of bone tissue and provoke skin disease (in severe cases, ulcerative lesions, swelling and necrosis of individual areas of the skin occur).
Receipt
Laboratory method for producing fluorine
The source for the production of fluorine is hydrogen fluoride HF, which is obtained mainly either by the action of sulfuric acid H2SO4 on fluorite CaF2, or by processing apatites and phosphorites.
Laboratory method
Laboratory conditions - fluorine can be obtained using chemical plants. Copper vessel 2, which has holes in the bottom, is placed in a copper vessel 1 filled with KF·3HF melt. A thick nickel anode is placed in vessel 2. The cathode is placed in vessel 1. Thus, during the electrolysis process, fluorine gas is released from tube 3, and hydrogen from tube 4. An important requirement is to ensure the tightness of the system; for this, plugs made of calcium fluoride with a lubricant of lead (II) oxide and glycerin are used.
In 1986, while preparing for a conference to celebrate the 100th anniversary of the discovery of fluorine, Karl Christe discovered a method for the purely chemical production of fluorine using the reaction in a hydrogen fluoride solution of K2MnF6 and SbF5 at 150 °C:
K2MnF6 + 2SbF5 → 2KSbF6 + MnF3 + ½F2 2K2MnF6 + 4SbF5 → 4KSbF6 + 2MnF3 + F2 Although this method has no practical application, it demonstrates that electrolysis is not necessary.
Industrial method
Industrial production of fluorine is carried out by electrolysis of a melt of acidic potassium fluoride KF·3HF (often with the addition of lithium fluoride), which is formed when the KF melt is saturated with hydrogen fluoride to a content of 40-41% HF. The electrolysis process is carried out at temperatures of about 100 °C in steel electrolyzers with a steel cathode and a carbon anode.
The benefits of fluoride for the human body
Fluoride has great benefits for the human body.
It performs many important functions:
- Inhibits the development of pathogenic microorganisms, preventing damage to tooth enamel by their metabolic products.
- Removes salts and heavy metals.
- Remineralizes enamel at the first stage of caries formation.
- Helps strengthen bone tissue.
- Normalizes phosphorus-calcium metabolism.
- Increases the body's resistance to radiation.
- Slows down the work of acid-forming bacteria.
- Responsible for the quality of hair and nails.
Chemical properties
The most active non-metal, it interacts violently with almost all substances (rare exceptions are fluoroplastics), and with most of them - with combustion and explosion. Contact of fluorine with hydrogen leads to ignition and explosion even at very low temperatures (down to −252°C). Even water and platinum burn in a fluorine atmosphere:
2F2 + 2H2O → 4HF + O2
Reactions in which fluorine is formally a reducing agent include the decomposition of higher fluorides, for example:
XeF8 → XeF6 + F2 MnF4 → MnF3 + 1/2 F2
Fluorine is also capable of oxidizing oxygen, forming oxygen fluoride OF2.
Fluorine. Properties of fluorine. Application of fluoride
Destruction and death. fluorine is translated from Greek . The name is associated with the history of its discovery. Dozens of scientists were injured or died trying to isolate the element whose existence Scheele first suggested. He obtained hydrofluoric acid, but was unable to extract a new substance from it - fluorium.
The name is associated with the mineral fluorite - the basis of hydrofluoric acid and the main source of fluorine . The Knox brothers from England and Gay-Lussac and Tenard from France also tried to obtain it by electrolysis. They died during the experiments.
Davy, who discovered sodium, potassium and calcium, contacted fluorium, was poisoned and became disabled. Afterwards, the scientific community renamed the element. But is it really that dangerous outside of chemical laboratories and why is it needed? We will answer these questions further.
Chemical and physical properties of fluorine
Fluorine occupies the 9th position in the periodic table. In nature, an element consists of a single stable nuclide. This is the name given to atoms whose life cycle is sufficient for observations and scientific research. The mass of a fluorine atom is 18.998. There are 2 atoms in a molecule.
Fluorine is the element with the highest electronegativity. The phenomenon is associated with the ability of an atom to connect with others and attract electrons to itself. Fluorine's index on the Pauling scale is 4. This contributes to the fame of the 9th element as the most active non-metal. In its normal state, it is a yellowish gas. It is toxic and has a pungent odor - something between the aromas of ozone and chlorine.
Fluorine is a substance with an abnormally low boiling point for gases - only 188 degrees Celsius. The remaining halogens, that is, typical non-metals from the 7th group of the periodic table, boil at high rates. This is due to the fact that they have a d-sublevel, which is responsible for one-and-a-half bonds. The fluorine molecule does not have one.
The activity of fluorine is expressed in the number and nature of possible reactions with other elements. Connection with most of them is accompanied by burning and explosions. In contact with hydrogen, a flame is generated even at low temperatures. Even water burns in a fluorine atmosphere. Moreover, in a chamber with a yellowish gas, platinum, the most inert and valuable element, ignites.
Fluorine compounds are impossible only with neon, argon and helium. All 3 gases are light and inert. Not from gases, diamond cannot be affected by fluorine. There are a number of elements with which reactions are possible only at elevated temperatures. Thus, the chlorine-fluorine interacts only at 200-250 degrees Celsius.
Application of fluoride
Teflon coatings cannot do without fluorine Their scientific name is tetrafluoroethylene. The compounds belong to the organic group and have non-stick properties. In essence, Teflon is a plastic, but unusually heavy. The density of water is 2 times higher - this is the reason for the excess weight of the coating and the dishes with it.
The fluorine content in Teflon is almost 70%. There is less of the element in freon, but this does not make the substance any less significant. The gas is used as a cooling agent. It is present in all refrigerators and air conditioners.
In the nuclear industry, fluorine has a connection with the process of separating uranium isotopes. Scientists say that if there were no 9th element, there would be no nuclear power plants. Not just any uranium serves as fuel for them, but only a few of its isotopes, in particular 235. Separation methods are designed for gases and volatile liquids.
But, uranium boils at 3500 degrees Celsius. It is unclear what materials for columns and centrifuges will withstand such heat. Fortunately, there is volatile uranium hexafluoride, which boils only at 57 degrees. It is from this that the metal fraction is isolated.
Fluorine oxidation , or more precisely, its oxidation of rocket fuel, is an important element of the aviation industry. It is not the gaseous element that is useful in it, but the liquid. In this state, fluorine turns bright yellow and is the most reactive.
In metallurgy, standard gas is used. The fluoride formula is being transformed. The element is included in the compound necessary to produce aluminum. It is produced by electrolysis. This is where hexafluoroaluminate is involved.
In optics, the magnesium-fluorine , that is, magnesium fluoride, is useful. It is transparent in the range of light waves from vacuum ultraviolet to infrared radiation. Here comes the connection to lenses and prisms for specialized optical instruments.
The 9th element was also noticed by doctors, in particular, dentists. They found 0.02% fluoride in teeth. Then it turned out that in regions where there is a shortage of the substance, the incidence of caries is higher.
is contained in water , from where it enters the body. In scarce areas, they began to artificially add the element to the water. The situation has improved. Therefore, a fluoride paste .
Fluoride in tooth enamel can cause fluorosis - darkening, spotting of tissues. This is a consequence of an overabundance of the element. Therefore, in regions with normal water composition, it is better to choose toothpaste without fluoride . It is also necessary to monitor its content in food products. There is even fluoridated milk. There is no need to enrich seafood; they already contain a lot of the 9th element.
Fluoride-free toothpaste is a choice related to the condition of the teeth. But in medicine, the element is needed not only in the field of dentistry. Fluoride preparations are prescribed for problems with the thyroid gland, for example, Graves' disease. In the fight against it, the fluorine-iodine .
Medicines with the 9th element are needed for those who have chronic diabetes. Glaucoma and cancer are also on the list of ailments that are treated with fluoride . As an oxygen substance, it is sometimes required for bronchial diseases and rheumatic diagnoses.
Fluorine extraction
Fluorine is extracted in the same way that helped to discover the element. After a series of deaths, one of the scientists managed not only to survive, but also to release a small amount of yellowish gas. The laurels went to Henri Moissan. The Frenchman was awarded the Nobel Prize for his discovery. It was issued in 1906.
Moissan used the electrolysis method. To avoid being poisoned by the fumes, the chemist carried out the reaction in a steel electrifier. This device is still used today. potassium fluoride is placed in it .
The process takes place at a temperature of 100 degrees Celsius. The cathode is made of steel. The anode in the installation is carbon. It is important to maintain the tightness of the system, because fluoride vapors are poisonous.
Laboratories purchase special plugs for tightness. Their composition: calcium-fluorine . The laboratory setup consists of two copper vessels. The first one is filled with melt, immersing the second one in it. The inner vessel has a hole in the bottom. A nickel anode passes through it.
The cathode is placed in the first vessel. Tubes extend from the device. Hydrogen is released from one, fluorine is released from the second. To maintain tightness, plugs and calcium fluoride alone are not enough. You also need lubrication. Its role is played by glycerin or lead oxide.
The laboratory method for obtaining the 9th element is used only for educational demonstrations. The technology has no practical application. However, its existence proves that it is possible to do without electrolysis. However, this is not necessary.
Fluorine price
There is no cost for fluoride as such. Prices are already set for products containing the 9th element of the periodic table. Toothpastes, for example, usually cost from 40 to 350 rubles. Medicines are also cheap and expensive. It all depends on the manufacturer and the availability of similar products from other companies on the market.
As for cost of fluoride , it can be seen to be high. The element is toxic. Handling it requires caution. Fluoride can be beneficial and even cure.
But for this you need to know a lot about the substance, predict its behavior and, of course, consult with specialists. Fluorine ranks 13th in terms of prevalence on Earth. The number itself, called the devil's dozen, forces you to be careful with the element.
Application in chemical activities (chemical industry)
Fluorine gas is used to produce:
uranium hexafluoride UF6 from UF4, used to separate uranium isotopes for the nuclear industry. chlorine trifluoride ClF3 - a fluorinating agent and a powerful oxidizer of rocket fuel sulfur hexafluoride SF6 - a gaseous insulator in the electrical industry of metal fluorides (for example, W and V), which have some useful properties freons - good refrigerants teflon - chemically inert polymers of sodium hexafluoroaluminate - for subsequent production aluminum by electrolysis of various fluorine compounds
Rocketry
Fluorine compounds are widely used in rocket technology as an oxidizer for rocket fuel.
Application in medicine
Fluorine compounds are widely used in medicine as blood substitutes.
What foods contain fluoride?
Fluorine, which is essential for the human body, is found in the following foods:
- walnuts and almonds;
- sea and freshwater fish;
- seafood (especially shrimp);
- vegetables (potatoes, pumpkin, onions, asparagus, avocado, Brussels sprouts, tomatoes, carrots, lettuce, spinach and other greens);
- eggs;
- fruits (grapefruit, apple, tangerine, plums);
- cereals (buckwheat, rice, corn, oats, bran, sprouted grains);
- beef and chicken;
- honey;
- pickles (pickles, tomatoes, cabbage);
- milk;
- black and green tea;
- red wine (the source here is the chemicals that fertilize grapes).
In order to receive the required amount of fluoride, it is not at all necessary to take pharmaceutical drugs. It is enough to include products from the above list in your menu, but also not to overdose on this element.
Biological and physiological role
Fluorine is a vital element for the body. In the human body, fluorine is mainly found in tooth enamel in the composition of fluorapatite - Ca5F(PO4)3. With insufficient (less than 0.5 mg/liter of drinking water) or excessive (more than 1 mg/liter) consumption of fluoride, the body can develop dental diseases: caries and fluorosis (mottling of enamel) and osteosarcoma, respectively.
A low fluorine content destroys enamel by leaching fluorine from fluorapatite to form hydroxoapatite, and vice versa.
To prevent caries, it is recommended to use toothpastes with fluoride additives or drink fluoridated water (up to a concentration of 1 mg/l), or use local applications of a 1-2% solution of sodium fluoride or stannous fluoride. Such actions can reduce the likelihood of tooth decay by 30-50%.
The maximum permissible concentration of bound fluorine in the air of industrial premises is 0.0005 mg/liter.
Daily fluoride intake
To avoid the negative consequences of fluoride deficiency and excess, you must adhere to the recommendations of experts regarding healthy doses of this substance. The human body’s need for the 9th element of the periodic table is covered by the daily fluorine norm established by doctors in accordance with age:
- infants should receive 0.01–0.5 mg/day of fluoride per day (this dose increases from lower to higher numbers from birth to one year); babies receive fluoride in water, breast milk, and baby food;
- from one year to 3 years – 0.7 mg / day;
- by 13 years of age the norm increases to 2 mg/day;
- young people 14–18 years old and women over 14 years old – 3 mg/day;
- adult men – 4 mg/day.
Additional Information
Fluorides Fluorine compounds in rocket technology Fluorine compounds in medicine Category:Fluorine compounds
Fluorine, Fluorum, F(9)
Fluorine (Fluorine, French and German Fluor) was obtained in a free state in 1886, but its compounds have been known for a long time and were widely used in metallurgy and glass production. The first mention of fluorite (CaP) under the name fluorspar (Fliisspat) dates back to the 16th century. One of the works attributed to the legendary Vasily Valentin mentions stones painted in various colors - flux (Fliisse from the Latin fluere - to flow, pour), which were used as fluxes in the smelting of metals. Agricola and Libavius write about this. The latter introduces special names for this flux - fluorspar (Flusspat) and mineral fluors. Many authors of chemical and technical works of the 17th and 18th centuries. describe different types of fluorspar. In Russia these stones were called fin, spalt, spat; Lomonosov classified these stones as selenites and called them spar or flux (crystal flux). Russian craftsmen, as well as collectors of mineral collections (for example, in the 18th century, Prince P.F. Golitsyn) knew that some types of spar when heated (for example, in hot water) glow in the dark. However, Leibniz, in his history of phosphorus (1710), mentions thermophosphorus (Thermophosphorus) in this regard.
Apparently, chemists and artisan chemists became acquainted with hydrofluoric acid no later than the 17th century. In 1670, the Nuremberg artisan Schwanhard used fluorspar mixed with sulfuric acid to etch patterns on glass goblets. However, at that time the nature of fluorspar and hydrofluoric acid was completely unknown. It was believed, for example, that silicic acid had a pickling effect in the Schwanhard process. This erroneous opinion was eliminated by Scheele, who proved that when fluorspar reacts with sulfuric acid, silicic acid is obtained as a result of the corrosion of a glass retort by the resulting hydrofluoric acid. In addition, Scheele established (1771) that fluorspar is a combination of calcareous earth with a special acid, which was called “Swedish acid”.
Lavoisier recognized the hydrofluoric acid radical as a simple body and included it in his table of simple bodies. Hydrofluoric acid was obtained in more or less pure form in 1809. Gay-Lussac and Thénard by distilling fluorspar with sulfuric acid in a lead or silver retort. During this operation, both researchers were poisoned. The true nature of hydrofluoric acid was established in 1810 by Ampere. He rejected Lavoisier's opinion that hydrofluoric acid should contain oxygen, and proved the analogy of this acid with hydrochloric acid. Ampere reported his findings to Davy, who had recently established the elemental nature of chlorine. Davy completely agreed with Ampere's arguments and spent a lot of effort on obtaining free fluorine by electrolysis of hydrofluoric acid and other ways. Taking into account the strong corrosive effect of hydrofluoric acid on glass, as well as on plant and animal tissues, Ampere proposed calling the element contained in it fluorine (Greek - destruction, death, pestilence, plague, etc.). However, Davy did not accept this name and proposed another - Fluorine, by analogy with the then name of chlorine - Chlorine, both names are still used in English. The name given by Ampere has been preserved in Russian.
Numerous attempts to isolate free fluorine in the 19th century. did not lead to successful results. Only in 1886 did Moissan manage to do this and obtain free fluorine in the form of a yellow-green gas. Since fluorine is an unusually aggressive gas, Moissan had to overcome many difficulties before he found a material suitable for equipment in experiments with fluorine. The U-tube for electrolysis of hydrofluoric acid at 55°C (cooled with liquid methyl chloride) was made of platinum with fluorspar plugs. After the chemical and physical properties of free fluorine were studied, it found wide application. Now fluorine is one of the most important components in the synthesis of a wide range of organofluorine substances. In Russian literature of the early 19th century. fluorine was called differently: hydrofluoric acid base, fluorin (Dvigubsky, 1824), fluoricity (Iovsky), fluor (Shcheglov, 1830), fluor, fluorine, fluoride. Hess introduced the name fluorine in 1831.
Harm of fluoride to the body
Fluoride can bring both benefits and harm to the human body. This substance is very dangerous in overdose. Fluoride takes a long time to be eliminated and accumulates not only in bones and teeth, but also in the pineal gland, which is located in the middle of the brain. In this case, its impact on human health can be very serious.
The issue of adding fluoride to municipal water, as well as various drinks, foods and toothpaste, is quite acute. In some countries (not Russia), synthetic fluorine compounds are added: fluorosilicic acid, sodium fluorosilicate, sodium fluoride.
Since such compounds are waste products from the phosphate, aluminum and even nuclear industries, their health hazards are obvious.
Sodium fluoride is the most harmful to health. Scientists claim that this substance reduces a person’s cognitive abilities (impairs speech, thinking ability and memory). Information about this became known to the general public only 10 years ago. Until this moment, the results of the study on the dangers of fluoride were classified.
Scientists have proven that long-term use of sodium fluoride causes the following disturbances in the normal functioning of the body:
- Malfunctions of the thyroid gland up to extensive disruption of the endocrine system.
- Genetic failures at the DNA level.
- Oncological diseases.
- Decreased melatonin production (which results in various sleep disorders).
- Neurological disorders (difficulties with focusing, memory, etc.).
- Alzheimer's disease.
Synthetic fluoride compounds have been used in the food industry since the 1940s. There are suggestions that sodium fluoride was used for the purpose of mass mind control in Nazi Germany, the USA and the Soviet Union.
Naturally occurring fluoride is part of the earth's crust, so a small amount of this substance in natural water is acceptable. Some fluorides are present in plants and animal tissues. However, even natural fluoride is a toxic substance for humans, even more dangerous than lead. Just 2-5 g is a lethal dose.
Fluoride Prevalence
Fluorine ranks 24th in abundance in the Universe. Its presence in space is assessed as low. For comparison, the remaining elements of the group can occur up to 20 times more. If you take the ratio, the amount of fluorine is around 400 parts per billion. Such a low concentration is due to the fact that the processes of stellar nucleosynthesis occur without the presence of fluorine. Any fluorine atoms formed have a high nuclear cross section, which later allows them to fuse with hydrogen or helium to concentrate oxygen or neon.
As for the Earth, fluorine is the thirteenth most abundant in the earth's crust. By weight, the ratio of parts varies around the figure of 400 parts per million. Fluorine, as a pure substance, does not occur in nature. Instead, it occurs as fluoride minerals. The most common of these is fluorite (CaF2). There are more than 9 thousand deposits of this mineral on our planet. The largest suppliers of fluorite are China and Mexico. Two other fluorine-containing minerals are fluoropathite, topaz and cryolite.
Read: Carbon as a chemical element on the periodic table
Industrial Application
In its atomic and molecular states, fluorine is used for plasma etching in the production of semiconductors, flat panel displays and microelectromechanical systems. Hydrofluoric acid is used for etching glass in lamps and other products.
Along with some of its compounds, fluorine is an important component in the production of pharmaceuticals, agrochemicals, fuels and lubricants and textiles. The chemical element is necessary for the production of halogenated alkanes (halons), which in turn were widely used in air conditioning and refrigeration systems. This use of chlorofluorocarbons was later banned because they contribute to the destruction of the ozone layer in the upper atmosphere.
Sulfur hexafluoride is an extremely inert, non-toxic gas classified as a greenhouse gas. Without fluorine, low-friction plastics such as Teflon cannot be produced. Many anesthetics (eg, sevoflurane, desflurane, and isoflurane) are hydrofluorocarbon derivatives. Sodium hexafluoroaluminate (cryolite) is used in the electrolysis of aluminum.
Fluoride compounds, including NaF, are used in toothpastes to prevent tooth decay. These substances are added to municipal water supplies to fluoridate the water, but the practice is considered controversial due to its effects on human health. At higher concentrations, NaF is used as an insecticide, especially to control cockroaches.
In the past, fluorides were used to lower the melting point of metals and ores and increase their fluidity. Fluorine is an important component in the production of uranium hexafluoride, which is used to separate its isotopes. 18F, a radioactive isotope with a half-life of 110 minutes, emits positrons and is often used in medical positron emission tomography.